About Us
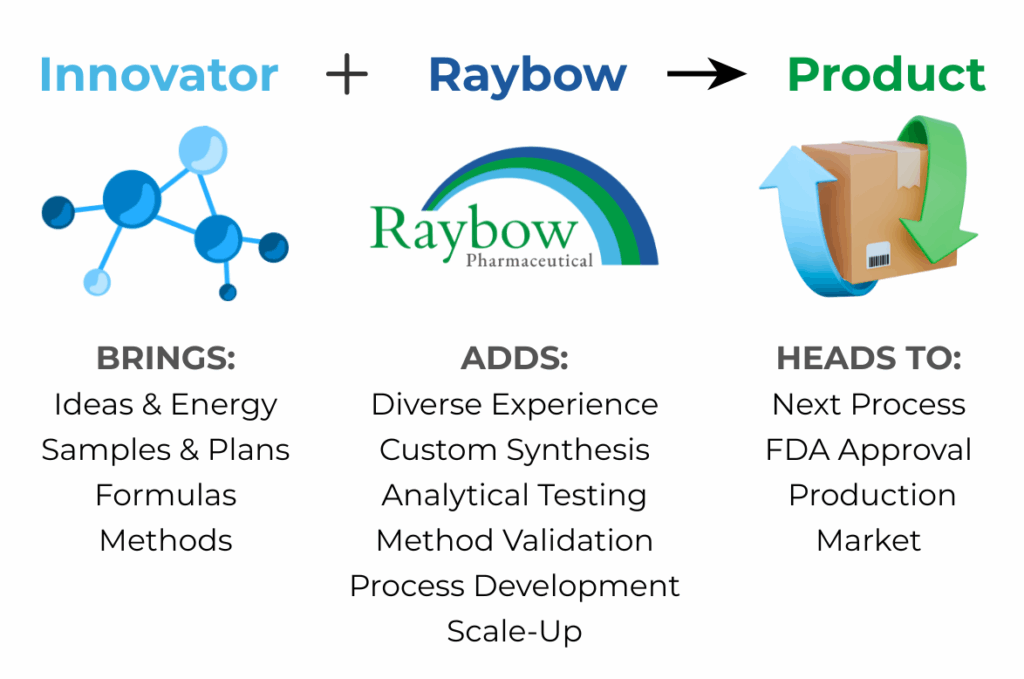
Raybow USA, located in Brevard, North Carolina, focuses on cGMP Pharmaceutical and Specialty Chemical Synthesis and Analytical Services in multiple markets. View sample service packages on the Services page. Learn more about the company and its pharmaceutical and specialty chemical capabilities on the At a Glance page.
Experience
25+ years in North Carolina
Providing multi-Kg scale commercial API from Brevard facilities
Expertise
PhD Chemists with diverse backgrounds work in teams to move your project forward
Synthetic and Analytical Labs collaborate to ensure focused progress
Efficiency
Experience project management team
Flexible contract formats
Cost competitive delivery
Excellence
FDA inspected facilities
Repeatedly passed strict EHS audits from multinational pharma companies
Raybow USA is part of the Jiuzhou Pharma global CDMO which focuses on R&D and green chemical manufacturing, with facilities and chemists in North America, Europe and China.
Raybow USA customers have seamless access to international experts and facilities.