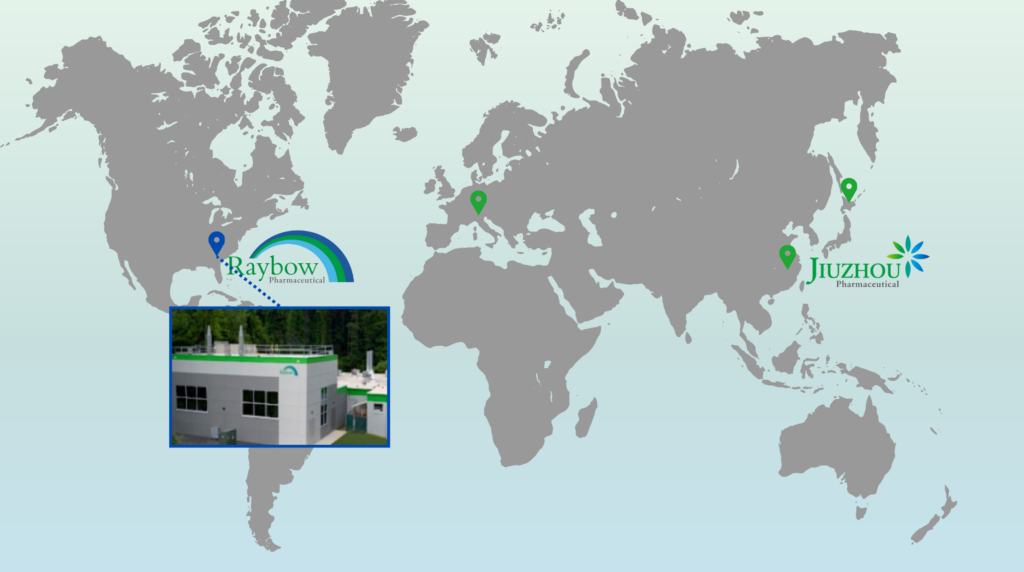
Brevard, NC, USA
Raybow USA in Brevard, North Carolina, formerly named PharmAgra Labs, was acquired by Jiuzhou Pharma in November 2019.
The Brevard site has a 25+ year history of providing contract R&D in organic and medicinal chemistry. The 11,400 square-foot facility assists clients with all phases of the discovery-development-clinical process.
Raybow USA provides customers with direct access to global resources through other Jiuzhou Pharma R&D and manufacturing sites.
Raybow USA, Inc.
158 McLean Road
Brevard, NC 28712
+1.828.884.8656